EN | JPN | FR

Creating vision through global research across 50+ countries
When your ophthalmic product shows promise, let's prove it., increased sensitivity. reduced variability..

35+ Indications 100% Ophthalmology

Our data solutions deliver precision and performance.
The preferred clinical research partner of ophthalmic innovators around the world..

Preclinical
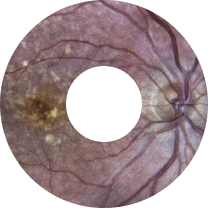
Retina, Glaucoma, Refractive & CED
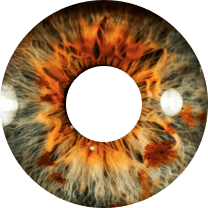
Dry Eye & Ocular Surface
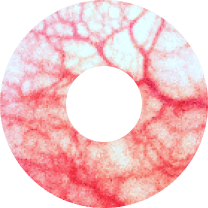
Allergy & Blepharitis

Medical Device
With 85+ approvals to date, whatever your challenge, we’re here to ensure success.

Discover how we make work better for our workforce
Ora, Inc websites use cookies. By continuing to browse the site you are agreeing to our use of cookies. For more details about cookies and their use, please see our Privacy Policy .
Ophthalmology CRO
Overcome complexities of ophthalmology trials and accelerate the path to approval with Medpace experts
Accelerating Complex Ophthalmology Clinical Trials with Specialized Expertise
With continuing innovations and advances in ophthalmology drug development, ophthalmology clinical trials are becoming increasingly complex. These trials require a clinical research organization (CRO) partner with innovative patient enrollment capabilities, strategic study design and execution, and cell, gene and RNA based therapy experience.
Accelerate your path to approval with guidance from our team of ophthalmology-focused medical, operational, and regulatory experts. Our extensive experience, world-class expertise, and consistent track record of success ensure the flexibility to adapt to the unique needs of each ophthalmology trial, overcome complex challenges, and keep trials on time and on budget.
Making the Complex Seamless ® in Global Ophthalmology Clinical Trials
Drive the successful execution of ophthalmology clinical trials through a partner with strong communication, coupled with early planning and experience collaborating with global regulatory authorities. Sponsors from emerging biotechs to global pharmaceutical companies have trusted Medpace for over 30 years to lead their ophthalmology development across a wide range of indications, including but not limited to Age-Related Macular Degeneration, Inherited Retinal Disease, Cataracts, Glaucoma, Cornea, Diabetic Macular Edema, and Natural History Studies.
Ophthalmology CRO Capabilities
Our medical, operations, and regulatory staff understand the complexities of ophthalmology trials from the perspective of the Sponsor, the clinical investigator, the scientific leader, and the reviewer at the regulatory agencies; we bring these perspectives to each clinical trial that we conduct to help keep trials running efficiently and according to protocol.
By partnering with a global CRO with a cross-functional collaborative model , the Medpace ophthalmology team can support your clinical trials through:
- Providing comprehensive support via our dedicated global regulatory submissions and global regulatory affairs teams
- Extensive experience partnering with the Sponsor’s choice of vendor for ophthalmology imaging to support each trial’s unique needs
- CAP-accredited central laboratory services for safety testing, PK/PD, biomarkers, and genomic testing; ensure the perfect harmonization of global data through wholly-owned and purpose-built laboratories with state-of-the-art infrastructure

Gene Therapy Research in Ophthalmology Clinical Trials
Executing ophthalmology gene therapy clinical trials is complex due to challenges including study procedures and assessments, site training, and sample storage. Our expertise and lessons learned in gene therapy ophthalmology clinical trials allow us to provide guidance to help Sponsors successfully navigate common complexities. Ensure successful patient recruitment and retention, effective investigator training, and reduce trial risks through our in-depth experience in gene therapy research across multiple therapeutic areas with both subretinal and intravitreal delivery.

Proactive Patient Recruitment & Retention
The success of clinical trials is wholly dependent on the success of sites and the engagement of patients. Our proprietary IntelliPACE ® feasibility strategy is informed by a multitude of internal and external sources to provide a highly customized, informed recruitment strategy for each study. Accelerate patient enrollment with strategic guidance from our dedicated, in-house patient recruitment and retention teams who are experienced in recruiting complex patient populations including pediatrics, geriatrics, and rare ocular diseases.
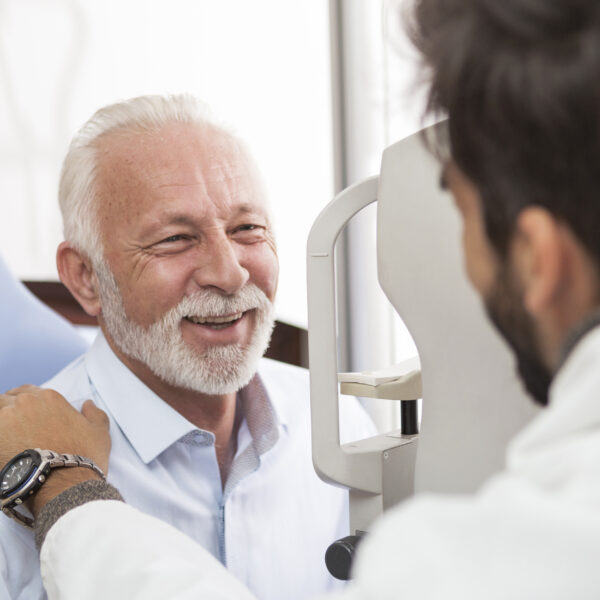
Strong, Long-Term Relationships
Medpace has built strong, long-term relationships with investigators, experienced sites, networks, and key opinion leaders specializing in ophthalmologic indications. The long-standing relationships combined with our experience help facilitate strategic site selection, start-up , and recruitment efforts translating into the successful and timely recruitment, conduct, and delivery of a study.

[Video] Medpace Ophthalmology – Meet Dr. Zaiba Malik
Ophthalmology clinical trial experts.
Our cross-functional, therapeutically trained teams include ophthalmology-specialized clinical trial managers and program coordinators that provide knowledgeable training to sites and operational leadership. In-house, board-certified ophthalmologists and gene therapy specialists add an additional layer of medical and scientific expertise and are embedded within the project team throughout the lifecycle of your trial. They provide leadership, consulting with Sponsors, along with training project teams and investigative sites to ensure our operational strategy is firmly aligned with your scientific and medical objectives.
“I see my role as an extension of the Sponsor and am available to guide and support them, especially for some smaller biotechs that often don’t have internal ophthalmologists on their teams.” Zaiba Malik, MD Medical Director, Medpace
Related Ophthalmology Insights

Fact Sheet: Ophthalmology
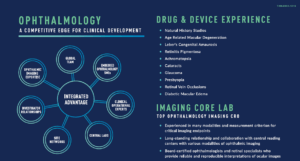
At-A-Glance: Ophthalmology
Innovations in ophthalmology clinical trials: medical, operational, regulatory and gene therapy considerations.

Operational Strategies for Success In Complex Ophthalmology Clinical Trials

Strategies to Accelerate Recruitment Projections in Global Phase II AMD Trials

Expert Insights: Interview with Dr. Marco Tangelder: New Developments in Ophthalmology
To give you the best possible experience, this site uses cookies and by continuing to use the site you agree that we can save them on your device.
- Schedule an Appointment
- Academic Advising
- Academic Life Coaching
- Academic Support & Skill Development
- Disability Support Services
- Peer Tutoring
- Multilingual Learning
- Choose a Major
- Assess Your Strengths & Skills
- Class Year Checklists
- Explore Careers
- What Can I Do with My Major?
- Arts & Media
- Communications
- Environmental
- Finance & Corporate
- Health & Wellness
- International
- Science, Technology, Engineering, and Math
- Social & Public Service
- First Generation
- International Students
- Students of Color
- Students with Disabilities
- Applied Methods and Research Experience
- APEX Fellowship
- Micro Fellowships
- Professional Development Mini Grants
- Sophomore Research
- Student Research
- Student Conference Travel
- Finding Internships
- Health Coach Program
- Research Opportunities
- Study Abroad & TREKs
- Job Simulations
- LinkedIn Learning
- Create a Resume
- Explore LinkedIn Learning
- Prepare for an Interview
- Network with Professionals
- Navigate the Application Process
- Seek Employment
- Wooster Connected Opportunities
- Apply to Graduate & Professional School
- Apply to Health Profession Programs
- Take a Gap Year
- Explore Fellowships & Service Programs
- Career Champions
- Faculty & Staff
- Parents & Families
- Academic Resource Center
- Career Planning
- Center for Entrepreneurship
- Experiential Learning & Community Engagement
- Global Engagement
- APEX Mission Statement
Stanford School of Medicine
Clinical research coordinator associate (hybrid) -ophthalmology.
- Share This: Share Clinical Research Coordinator Associate (Hybrid) -Ophthalmology on Facebook Share Clinical Research Coordinator Associate (Hybrid) -Ophthalmology on LinkedIn Share Clinical Research Coordinator Associate (Hybrid) -Ophthalmology on X
The Byers Eye Institute at Stanford University School of Medicine is seeking a Clinical Research Coordinator Associate to perform duties related to the coordination of clinical studies under the direction of Dr. Andrea Kossler, Associate Professor of Ophthalmology. Duties include coordination of moderately complex aspects of one or more clinical studies. The incumbent will work under close direction of the principal investigator and/or study coordinator/supervisor on studies related to Thyroid eye disease, among other diseases. More information can be found on our Clinical Trials website: https://med.stanford.edu/ophthalmology/research/clinical_trials.html.
Duties include:
- Serve as primary contact with research participants, sponsors, and regulatory agencies. Coordinate studies from startup through close-out.
- Determine eligibility of and gather consent from study participants according to protocol. Assist in developing recruitment strategies.
- Coordinate collection of study specimens and processing.
- Collect and manage patient and laboratory data for clinical research projects. Manage research project databases, develop flow sheets and other study related documents, and complete study documents/case report forms.
- Ensure compliance with research protocols, and review and audit case report forms for completion and accuracy with source documents. Prepare regulatory submissions, and ensure institutional Review Board renewals are completed.
- Assemble study kits for study visits, monitor scheduling of procedures and charges, coordinate documents, and attend monitoring meetings with sponsors, acting as primary contact.
- Monitor expenditures and adherence to study budgets and resolve billing issues in collaboration with finance and/or management staff.
- Interact with the principal investigator regularly, ensuring patient safety and adherence to proper study conduct.
- Ensure essential documentation and recording of patient and research data in appropriate files per institutional and regulatory requirements.
- Participate in monitor visits and regulatory audits.
- – Other duties may also be assigned
EDUCATION & EXPERIENCE (REQUIRED):
Two year college degree and two years related work experience or a Bachelor’s degree in a related field or an equivalent combination of related education and relevant experience.
KNOWLEDGE, SKILLS AND ABILITIES (REQUIRED):
· Strong interpersonal skills.
· Proficiency with Microsoft Office.
· Knowledge of medical terminology.
CERTIFICATIONS & LICENSES:
Society of Clinical Research Associates or Association of Clinical Research Professionals certification is preferred.
PHYSICAL REQUIREMENTS*:
· Frequently stand, walk, twist, bend, stoop, squat and use fine light/fine grasping.
· Occasionally sit, reach above shoulders, perform desk based computer tasks, use a telephone and write by hand, lift, carry, push, and pull objects that weigh up to 40 pounds.
· Rarely kneel, crawl, climb ladders, grasp forcefully, sort and file paperwork or parts, rarely lift, carry, push, and pull objects that weigh 40 pounds or more.
* – Consistent with its obligations under the law, the University will provide reasonable accommodation to any employee with a disability who requires accommodation to perform the essential functions of his or her job.
WORKING CONDITIONS:
Occasional evening and weekend hours.
The expected pay range for this position is $31.73 to $36.54 per hour.
Stanford University provides pay ranges representing its good faith estimate of what the university reasonably expects to pay for a position. The pay offered to a selected candidate will be determined based on factors such as (but not limited to) the scope and responsibilities of the position, the qualifications of the selected candidate, departmental budget availability, internal equity, geographic location and external market pay for comparable jobs.
At Stanford University, base pay represents only one aspect of the comprehensive rewards package. The Cardinal at Work website ( https:/lcardinalatwork.stanford.edu/benefits-rewards ) provides detailed information on Stanford’s extensive range of benefits and rewards offered to employees. Specifics about the rewards package for this position may be discussed during the hiring process.
USC Career Services is moving to Handshake. connectSC will not be available starting July 1, 2024. Learn more

- Faculty & Staff
- Parents & Families
- First-Generation Students
- International Students
- Job Seekers Who Were Formerly Incarcerated
- LGBTQ+ Students
- Student Accessibility
- Students of Color
- Undocumented & DACA Students
- Veteran Students
- Womxn Students
- Advertising, Marketing, and PR
- Architecture, Construction, and Design
- Business Management and Administration
- Computer Science and Information Technology
- Consulting and Finance
- Data Science, Statistics, and Artificial Intelligence
- Education and Research
- Engineering
- Entertainment, Media, and Communications
- Environment and Sustainability
- Government, Non-Profit, and Public Administration
- Health and Life Sciences, Biotech, and Pharmaceuticals
- International Opportunities
- Law, Policy, and Social Justice
- Performing and Visual Arts
- Career Development Process
- Appointments & Requests
- Explore Events
- Personal Finance
- Resumes/Cover Letters/Curriculum Vitaes
- Interview Preparation
- Offer Negotiation
- On-Campus Jobs
- On-Campus Recruiting
- Workplace Success
- Professional Development Advice
- eNewsletter
- Meet the Team
- School-Based Career Services
Stanford School of Medicine
Clinical research coordinator associate (onsite) -ophthalmology.
- Share This: Share Clinical Research Coordinator Associate (Onsite) -Ophthalmology on Facebook Share Clinical Research Coordinator Associate (Onsite) -Ophthalmology on LinkedIn Share Clinical Research Coordinator Associate (Onsite) -Ophthalmology on X
The Byers Eye Institute at Stanford University is seeking a Clinical Research Coordinator Associate to perform duties related to the coordination of clinical studies in the lab of Dr. Tawna Roberts ( https://med.stanford.edu/roberts-lab.html ). Supporting exciting research in vision development, visual neuroscience, amblyopia, strabismus, and vision disorders related brain injury, the CRCA will coordinate moderately complex aspects of one or more clinical studies. You will work under close direction of the principal investigator to advance the mission of excellence in patient care and cutting-edge clinical research sponsored by the National Institute of Health/National Eye Institute, Department of Defense, Pediatric Eye Disease Investigator Group, and another funding foundations.
As an organization that receives federal funding, Stanford University has a COVID-19 vaccination requirement that will apply to all university employees, including those working remotely in the United States and applicable subcontractors. To learn more about COVID policies and guidelines for Stanford University Staff, please visit https://healthalerts.stanford.edu/covid-19/ .
Duties include:
- Serve as primary contact with research participants, sponsors, and regulatory agencies. Coordinate studies from startup through close-out.
- Determine eligibility of and gather consent from study participants according to protocol. Assist in developing recruitment strategies.
- Coordinate collection of study specimens and processing.
- Collect and manage patient and laboratory data for clinical research projects. Manage research project databases, develop flow sheets and other study related documents, and complete study documents/case report forms.
- Ensure compliance with research protocols, and review and audit case report forms for completion and accuracy with source documents. Prepare regulatory submissions, and ensure institutional Review Board renewals are completed.
- Assemble study kits for study visits, monitor scheduling of procedures and charges, coordinate documents, and attend monitoring meetings with sponsors, acting as primary contact.
- Monitor expenditures and adherence to study budgets and resolve billing issues in collaboration with finance and/or management staff.
- Interact with the principal investigator regularly, ensuring patient safety and adherence to proper study conduct.
- Ensure essential documentation and recording of patient and research data in appropriate files per institutional and regulatory requirements.
- Participate in monitor visits and regulatory audits.
- * – Other duties may also be assigned
EDUCATION & EXPERIENCE (REQUIRED):
Two year college degree and two years related work experience or a Bachelor’s degree in a related field or an equivalent combination of related education and relevant experience.
KNOWLEDGE, SKILLS AND ABILITIES (REQUIRED):
- Strong interpersonal skills.
- Proficiency with Microsoft Office.
- Knowledge of medical terminology.
CERTIFICATIONS & LICENSES:
- Society of Clinical Research Associates or Association of Clinical Research Professionals certification is preferred.
PHYSICAL REQUIREMENTS*:
- Frequently stand, walk, twist, bend, stoop, squat and use fine light/fine grasping.
- Occasionally sit, reach above shoulders, perform desk based computer tasks, use a telephone and write by hand, lift, carry, push, and pull objects that weigh up to 40 pounds.
- Rarely kneel, crawl, climb ladders, grasp forcefully, sort and file paperwork or parts, rarely lift, carry, push, and pull objects that weigh 40 pounds or more.
- * – Consistent with its obligations under the law, the University will provide reasonable accommodation to any employee with a disability who requires accommodation to perform the essential functions of his or her job.
WORKING CONDITIONS:
- Occasional evening and weekend hours.
The expected pay range for this position is $27.88 to $36.54 per hour. Stanford University provides pay ranges representing its good faith estimate of what the university reasonably expects to pay for a position. The pay offered to a selected candidate will be determined based on factors such as (but not limited to) the scope and responsibilities of the position, the qualifications of the selected candidate, departmental budget availability, internal equity, geographic location and external market pay for comparable jobs. At Stanford University, base pay represents only one aspect of the comprehensive rewards package. The Cardinal at Work website (https:/lcardinalatwork.stanford.edu/benefits-rewards) provides detailed information on Stanford’s extensive range of benefits and rewards offered to employees. Specifics about the rewards package for this position may be discussed during the hiring process.
Consistent with its obligations under the law, the University will provide reasonable accommodations to applicants and employees with disabilities. Applicants requiring a reasonable accommodation for any part of the application or hiring process should contact Stanford University Human Resources at [email protected]. For all other inquiries, please submit a contact form. Stanford is an equal employment opportunity and affirmative action employer. All qualified applicants will receive consideration for employment without regard to race, color, religion, sex, sexual orientation, gender identity, national origin, disability, protected veteran status, or any other characteristic protected by law. Consistent with its obligations under the law, the University will provide reasonable accommodation to any employee with a disability who requires accommodation to perform the essential functions of the job.

- Search Menu
- Sign in through your institution
- Advance articles
- Author Guidelines
- Submission Site
- Open Access
- Why Submit?
- About Public Opinion Quarterly
- About the American Association for Public Opinion Research
- Editorial Board
- Advertising and Corporate Services
- Journals Career Network
- Self-Archiving Policy
- Dispatch Dates
- Journals on Oxford Academic
- Books on Oxford Academic

Article Contents
Anti-semitic attitudes of the mass public: estimates and explanations based on a survey of the moscow oblast.
- Article contents
- Figures & tables
- Supplementary Data
JAMES L. GIBSON, RAYMOND M. DUCH, ANTI-SEMITIC ATTITUDES OF THE MASS PUBLIC: ESTIMATES AND EXPLANATIONS BASED ON A SURVEY OF THE MOSCOW OBLAST, Public Opinion Quarterly , Volume 56, Issue 1, SPRING 1992, Pages 1–28, https://doi.org/10.1086/269293
- Permissions Icon Permissions
In this article we examine anti-Semitism as expressed by a sample of residents of the Moscow Oblast (Soviet Union). Based on a survey conducted in 1920, we begin by describing anti-Jewish prejudice and support for official discrimination against Jews. We discover a surprisingly low level of expressed anti-Semitism among these Soviet respondents and virtually no support for state policies that discriminate against Jews. At the same time, many of the conventional hypotheses predicting anti-Semitism are supported in the Soviet case. Anti-Semitism is concentrated among those with lower levels of education, those whose personal financial condition is deteriorating, and those who oppose further democratization of the Soviet Union. We do not take these findings as evidence that anti-Semitism is a trivial problem in the Soviet Union but, rather, suggest that efforts to combat anti-Jewish movements would likely receive considerable support from ordinary Soviet people.
Email alerts
Citing articles via.
- Recommend to your Library
Affiliations
- Online ISSN 1537-5331
- Copyright © 2024 American Association for Public Opinion Research
- About Oxford Academic
- Publish journals with us
- University press partners
- What we publish
- New features
- Open access
- Institutional account management
- Rights and permissions
- Get help with access
- Accessibility
- Advertising
- Media enquiries
- Oxford University Press
- Oxford Languages
- University of Oxford
Oxford University Press is a department of the University of Oxford. It furthers the University's objective of excellence in research, scholarship, and education by publishing worldwide
- Copyright © 2024 Oxford University Press
- Cookie settings
- Cookie policy
- Privacy policy
- Legal notice
This Feature Is Available To Subscribers Only
Sign In or Create an Account
This PDF is available to Subscribers Only
For full access to this pdf, sign in to an existing account, or purchase an annual subscription.
Ohio State nav bar
The Ohio State University
- BuckeyeLink
- Find People
- Search Ohio State
Promotion and Tenure Approvals 2024
This week brought many Ohio State faculty members a reason to celebrate as the Board of Trustees approved promotion and tenure recommendations for the Buckeyes listed below. Each has excelled in the areas of teaching, research and creative accomplishments, and service. Interim Executive Vice President and Karla Zadnik and the Office of Academic Affairs is proud to recognize these achievements and contributions to the university.
All promotions became effective on May 16, 2024.

College of Arts and Sciences – Division of Art and Humanities
Promotion to Professor:
Breyfogle, Nicholas, History Burry, Alexander, Slavic and East European Languages and Cultures Duenas, Alcira, History, Newark Hawkins, Tom, Classics Hoffmann, Benjamin, French and Italian Judd, Robin, History Lockett, Leslie, English Low, John, Comparative Studies, Newark Rush, George, Art Sims, Andrea, Linguistics Skinner, Ryan, School of Music Steinmetz, Brad, Theatre, Film, and Media Arts Teston, Christa, English Wallace, Shawn, School of Music Weiner, Isaac, Comparative Studies Whittington, Karl, History of Art Winant, Carmen, Art
Promotion to Associate Professor with Tenure:
Kantor, Jamison, English, Mansfield Lewis, Matt, Design Perez, Ashley, Comparative Studies Rask, Katie, Classics, Marion Toth, Lucille, French and Italian, Newark
College of Arts and Sciences – Division of Natural and Mathematical Sciences
Burd, Christin, Molecular Genetics Griffith, W. Ashley, School of Earth Sciences Johnson, Niles, Mathematics, Newark Katz, Eric, Mathematics Koenig, Kenneth, Mathematics Landsman, Alexandra, Physics Lindert, Steffen, Chemistry and Biochemistry Lu, Yuanming, Physics Moortgat, Joachim, School of Earth Sciences Nakanishi, Kotaro, Chemistry and Biochemistry Patrikis, Stefan, Mathematics Penneys, David, Mathematics Sawyer, Audrey, School of Earth Sciences
Asta, Dena, Statistics Gautam, Sachin, Mathematics Jarzyna, Marta, Evolution, Ecology and Organismal Biology Ju, Kou-San, Microbiology Krishnan, Sanjeevi, Mathematics Leonard-Pingel, Jill, School of Earth Sciences, Newark Park, Jennifer, Mathematics Sivakoff, Frances, Evolution, Ecology and Organismal Biology, Marion Skinner, Brian, Physics Wang, Ji, Astronomy
College of Arts and Sciences – Division of Social and Behavioral Sciences
Carroll, Patrick, Psychology, Lima Colen, Cynthia, Sociology DeAndrea, David, School of Communication Harnish, Stacy, Speech and Hearing Science Lopez, Steven, Sociology Madsen, Kenneth, Geography, Newark Moyer-Guse, Emily, School of Communication Patson, Nikole, Psychology, Marion Price-Spratlen, Townsand, Sociology Tompkins, Virginia, Psychology, Lima Way, Baldwin, Psychology
Dutta, Madhumita, Geography Lin, Erin, Political Science Ritchie, Melinda, Political Science Saunders-Hastings, Emma, Political Science
College of Arts and Sciences – Clinical
Promotion to Associate Professor-Clinical:
Chapman, Bridget, Speech and Hearing Science
Max M. Fisher College of Business
Dumas, Tracy, Management & Human Resources Goodman, Joseph, Marketing & Logistics Wan, Sean, Marketing & Logistics
Lee, Hun, Management & Human Resources
Draper, John, Operations and Business Analytics
College of Dentistry
Beetstra, Steve Cottle, James Messina, Matthew Novopoltseva, Irina Wenzel, Mark
College of Education and Human Ecology
Brock, Matthew, Educational Studies Troyan, Francis, Teaching and Learning Yilmazer, Tansel, Human Sciences
Ansari, Arya, Human Sciences Harrison, Dorian, Teaching and Learning, Newark Landers, Ashley, Human Sciences
Fast, Danene, Teaching and Learning
College of Engineering
Brunelli, Nick, Chemical and Biomolecular Engineering Conroy, Maria, Knowlton School of Architecture, Gallego-Perez, Daniel, Biomedical Engineering Ghazisaeidi, Maryam, Materials Science and Engineering Niezgoda, Steve, Materials Science and Engineering Prakash, Shaurya, Mechanical and Aerospace Engineering Sertel, Kubilay, Electrical and Computer Engineering Stewart, Christopher, Computer Science and Engineering
Bigham, Ashley, Knowlton School of Architecture Burris, Lisa, Civil, Environmental, and Geodetic Engineering Davanloo, Sam, Integrated Systems Engineering Herrmann, Erik, Knowlton School of Architecture Liu, Jia (Kevin), Electrical and Computer Engineering Liu, Xun, Materials Science and Engineering Steiner, Halina, Knowlton School of Architecture
Promotion to Professor-Clinical:
Nocera, Tanya, Biomedical Engineering
Tahmina, Qudsia, Electrical and Computer Engineering, Marion
College of Food, Agricultural, and Environmental Sciences
Adams, Ingrid, Extension Hand, Francesca, Plant Pathology Hedrick, Jason, Extension Johnson, Reed, Entomology Romich, Eric, Extension Slot, Jason, Plant Pathology Zoller, Christopher, Extension
Bowling, Amanda, Agricultural Communication, Education, and Leadership Enger, Benjamin, Animal Sciences Garcia Guerra, Alvaro, Animal Sciences Hamilton, Matthew, School of Environment and Natural Resources Jenkins, Margaret, Extension Khanal, Sami, Food, Agricultural, and Biological Engineering Niewoehner-Green, Jera, Agricultural Communication, Education, and Leadership Richer, Eric, Extension West, Travis, Extension Winston, Ryan, Food, Agricultural, and Biological Engineering
Klooster, Wendy, Horticulture and Crop Science
Michael E. Moritz College of Law
Conway, Olwyn Roser-Jones, Courtlyn
College of Medicine
Bell, Charles, Biological Chemistry and Pharmacology Gu, Chen, Biological Chemistry and Pharmacology Gumina, Richard, Internal Medicine Karuppaiyah, Selvendiran, Obstetrics and Gynecology Kolipaka, Arunark, Radiology O'Brien, Sarah, Pediatrics Onate, James, School of Health and Rehabilitation Sciences Roychowdhury, Sameek, Internal Medicine Singh, Harpreet, Physiology and Cell Biology Slaughter, Jonathan, Pediatrics Spees, Colleen, School of Health and Rehabilitation Sciences Stevens, John, Pediatrics Zhao, Kai, Otolaryngology Zhu, Jian, Pathology
Choe, Hannah, Internal Medicine Eisfeld, Ann-Kathrin, Internal Medicine Jackson, Jamie, Pediatrics Juckett, Lisa, School of Health and Rehabilitation Sciences Krok-Schoen, Jessica, School of Health and Rehabilitation Sciences Mace, Thomas, Internal Medicine Meyer, Kathrin, Pediatrics Plascak, Jesse, Internal Medicine Tackett, Alayna, Internal Medicine Tedeschi, Andrea, Neuroscience Venkatesh, Kartik, Obstetrics and Gynecology Wisler, Jon, Surgery Zhao, Mingtao, Pediatrics
Promotion to Associate Professor without Tenure:
Cherian, Mathew, Internal Medicine Schenk, Austin, Surgery
Bajwa, Rajinder, Pediatrics Baughcum, Amy, Pediatrics Blakaj, Dukagjin, Radiation Oncology Carr, David, Dermatology Cuff, Steven, Pediatrics Denny, Sarah, Pediatrics Kang, Stephen, Otolaryngology Lampert, Brent, Internal Medicine Letson, Megan, Pediatrics Lilly, Scott, Internal Medicine Lind, Meredith, Otolaryngology McGwire, Gerd, Pediatrics McLaughlin, Douglas, Pediatrics Naguib, Aymen, Anesthesiology Nahata, Leena, Pediatrics O'Brien, Nicole, Pediatrics Pearson, Gregory, Plastic and Reconstructive Surgery Powers, Ciaran, Neurological Surgery Prevedello, Luciano, Radiology Price, Jonathan, Pediatrics Shah, Zarine, Radiology Tozbikian, Gary, Pathology Wesolowski, Robert, Internal Medicine Witman, Patricia, Pediatrics Yardley, Heather, Pediatrics Yates, Andrew, Pediatric
Ackermann, Wiebke, Anesthesiology Almaani, Salem, Internal Medicine Bajwa, Rajinder, Pediatrics Baughcum, Amy, Pediatrics Beyer, Sasha, Radiation Oncology Bhateja, Priyanka, Internal Medicine Blakaj, Dukagjin, Radiation Oncology Bond, David, Internal Medicine Bouchard, Traci, Pediatrics Breckenridge, Mary, Internal Medicine Carr, David, Dermatology Cetnar, Ashley, Radiation Oncology Chaparro, Juan, Pediatrics Chen, Sonja, Pathology Ciciora, Steven, Pediatrics Cooper, Avraham, Internal Medicine Crichton, Kristin, Pediatrics Cuff, Steven, Pediatrics Dason, Shawn, Urology Denny, Sarah, Pediatrics Dudley, Samuel, Pediatrics Dulmage, Brittany, Dermatology Ehrman, Sarah, Internal Medicine El-Ferzli, George, Pediatrics Farrell, Matthew, Family and Community Medicine Frazier, Warren, Pediatrics Fried, Martin, Internal Medicine Ganapathi, Asvin, Surgery Gathof, Andrew, Emergency Medicine Grossbach, Andrew, Neurological Surgery Haisley, Kelly, Surgery Halaweish, Ihab, Surgery Hostutler, Cody, Pediatrics Kang, Stephen, Otolaryngology Kneile, Jeffrey, Pathology Kobalka, Peter, Pathology Krivchenia, Katelyn, Pediatrics Lampert, Brent, Internal Medicine Letson, Megan, Pediatrics Li, Jieli "Shirley", Pathology Liao, Nancy, Pediatrics Lilly, Scott, Internal Medicine Lind, Meredith, Otolaryngology Lloyd, Julia, Pediatrics Makary, Mina, Radiology McGwire, Gerd, Pediatrics McLaughlin, Douglas, Pediatrics Melvin, Jennifer, Pediatrics Moreno, Jose, Psychiatry and Behavioral Health Mori, Mari, Pediatrics Mosser-Goldfarb, Joy, Pediatrics Mynatt, Irene, Emergency Medicine Naguib, Aymen, Anesthesiology Nahata, Leena, Pediatrics Nguyen, Christopher, Psychiatry and Behavioral Health Nolan, Larry, Family and Community Medicine Noon, Kristen, Anesthesiology O'Brien, Nicole, Pediatrics Obrynba, Kathryn, Pediatrics Paul, Jisna, Internal Medicine Pearson, Gregory, Plastic and Reconstructive Surgery Powers, Ciaran, Neurological Surgery Price, Jonathan, Pediatrics Rust, Laura, Pediatrics Schaffernocker, Troy, Internal Medicine Shah, Zarine, Radiology Siddiqui, Irmeen, Pathology Smajlovic, Amina, Pediatrics Sourial, Michael, Urology Stearns, Walter, Psychiatry and Behavioral Health Streby, Keri, Pediatrics Stukus, Kristin, Pediatrics Sullivan, Anne, Orthopaedics Swanson, Melissa, Pediatrics Tang, Bingfeng, Radiology The Ohio State University May 15, 2024 Board of Trustees Thomas, Diana, Pathology Thompson, Gina, Pediatrics Tozbikian, Gary, Pathology Wang, Huanyu, Pathology Wesolowski, Robert, Internal Medicine Witman, Patricia, Pediatrics Xu, David, Neurological Surgery Yang, Yuanquan, Internal Medicine Yardley, Heather, Pediatrics Yates, Andrew, Pediatrics Youssef, Patrick, Neurological Surgery
Promotion to Research Professor
Fontanella, Cynthia, Psychiatry and Behavioral Health
Promotion to Research Associate Professor:
Mansfield, Julie, School of Health and Rehabilitation Sciences
Tenure (at current rank of associate professor):
Farkas, Laszlo, Internal Medicine Hays, John, Internal Medicine Kerur, Nagaraj, Ophthalmology and Visual Sciences
College of Nursing
O'Mathuna, Donal
Wright, Kathy
College of Optometry
Choi, Stacey Doble, Nathan
College of Pharmacy
Cocucci, Ema Coss, Christopher Hu, Shuiying
John Glenn College of Public Affairs
Clark, Jill
LePere-Schloop, Megan
College of Public Health
Andridge, Rebecca Pennell, Michael
Hogan, Tory Nemeth, Julianna Ricks, JaNelle Schnell, Patrick
College of Social Work
Boyd, Donte Oh, Sehun
College of Veterinary Medicine
Bowman, Andrew, Veterinary Preventive Medicine Marsh, Antoinette, Veterinary Preventive Medicine
Berrian, Amanda, Veterinary Preventive Medicine Hale, Vanessa, Veterinary Preventive Medicine
University Libraries
Frazier, Mara Hammons, Jane
- Yekaterinburg
- Novosibirsk
- Vladivostok

- Tours to Russia
- Practicalities
- Russia in Lists
Rusmania • Deep into Russia
Out of the Centre
Savvino-storozhevsky monastery and museum.

Zvenigorod's most famous sight is the Savvino-Storozhevsky Monastery, which was founded in 1398 by the monk Savva from the Troitse-Sergieva Lavra, at the invitation and with the support of Prince Yury Dmitrievich of Zvenigorod. Savva was later canonised as St Sabbas (Savva) of Storozhev. The monastery late flourished under the reign of Tsar Alexis, who chose the monastery as his family church and often went on pilgrimage there and made lots of donations to it. Most of the monastery’s buildings date from this time. The monastery is heavily fortified with thick walls and six towers, the most impressive of which is the Krasny Tower which also serves as the eastern entrance. The monastery was closed in 1918 and only reopened in 1995. In 1998 Patriarch Alexius II took part in a service to return the relics of St Sabbas to the monastery. Today the monastery has the status of a stauropegic monastery, which is second in status to a lavra. In addition to being a working monastery, it also holds the Zvenigorod Historical, Architectural and Art Museum.
Belfry and Neighbouring Churches

Located near the main entrance is the monastery's belfry which is perhaps the calling card of the monastery due to its uniqueness. It was built in the 1650s and the St Sergius of Radonezh’s Church was opened on the middle tier in the mid-17th century, although it was originally dedicated to the Trinity. The belfry's 35-tonne Great Bladgovestny Bell fell in 1941 and was only restored and returned in 2003. Attached to the belfry is a large refectory and the Transfiguration Church, both of which were built on the orders of Tsar Alexis in the 1650s.

To the left of the belfry is another, smaller, refectory which is attached to the Trinity Gate-Church, which was also constructed in the 1650s on the orders of Tsar Alexis who made it his own family church. The church is elaborately decorated with colourful trims and underneath the archway is a beautiful 19th century fresco.
Nativity of Virgin Mary Cathedral

The Nativity of Virgin Mary Cathedral is the oldest building in the monastery and among the oldest buildings in the Moscow Region. It was built between 1404 and 1405 during the lifetime of St Sabbas and using the funds of Prince Yury of Zvenigorod. The white-stone cathedral is a standard four-pillar design with a single golden dome. After the death of St Sabbas he was interred in the cathedral and a new altar dedicated to him was added.

Under the reign of Tsar Alexis the cathedral was decorated with frescoes by Stepan Ryazanets, some of which remain today. Tsar Alexis also presented the cathedral with a five-tier iconostasis, the top row of icons have been preserved.
Tsaritsa's Chambers

The Nativity of Virgin Mary Cathedral is located between the Tsaritsa's Chambers of the left and the Palace of Tsar Alexis on the right. The Tsaritsa's Chambers were built in the mid-17th century for the wife of Tsar Alexey - Tsaritsa Maria Ilinichna Miloskavskaya. The design of the building is influenced by the ancient Russian architectural style. Is prettier than the Tsar's chambers opposite, being red in colour with elaborately decorated window frames and entrance.

At present the Tsaritsa's Chambers houses the Zvenigorod Historical, Architectural and Art Museum. Among its displays is an accurate recreation of the interior of a noble lady's chambers including furniture, decorations and a decorated tiled oven, and an exhibition on the history of Zvenigorod and the monastery.
Palace of Tsar Alexis

The Palace of Tsar Alexis was built in the 1650s and is now one of the best surviving examples of non-religious architecture of that era. It was built especially for Tsar Alexis who often visited the monastery on religious pilgrimages. Its most striking feature is its pretty row of nine chimney spouts which resemble towers.

Plan your next trip to Russia
Ready-to-book tours.
Your holiday in Russia starts here. Choose and book your tour to Russia.
The Unique Burial of a Child of Early Scythian Time at the Cemetery of Saryg-Bulun (Tuva)
<< Previous page
Pages: 379-406
In 1988, the Tuvan Archaeological Expedition (led by M. E. Kilunovskaya and V. A. Semenov) discovered a unique burial of the early Iron Age at Saryg-Bulun in Central Tuva. There are two burial mounds of the Aldy-Bel culture dated by 7th century BC. Within the barrows, which adjoined one another, forming a figure-of-eight, there were discovered 7 burials, from which a representative collection of artifacts was recovered. Burial 5 was the most unique, it was found in a coffin made of a larch trunk, with a tightly closed lid. Due to the preservative properties of larch and lack of air access, the coffin contained a well-preserved mummy of a child with an accompanying set of grave goods. The interred individual retained the skin on his face and had a leather headdress painted with red pigment and a coat, sewn from jerboa fur. The coat was belted with a leather belt with bronze ornaments and buckles. Besides that, a leather quiver with arrows with the shafts decorated with painted ornaments, fully preserved battle pick and a bow were buried in the coffin. Unexpectedly, the full-genomic analysis, showed that the individual was female. This fact opens a new aspect in the study of the social history of the Scythian society and perhaps brings us back to the myth of the Amazons, discussed by Herodotus. Of course, this discovery is unique in its preservation for the Scythian culture of Tuva and requires careful study and conservation.
Keywords: Tuva, Early Iron Age, early Scythian period, Aldy-Bel culture, barrow, burial in the coffin, mummy, full genome sequencing, aDNA
Information about authors: Marina Kilunovskaya (Saint Petersburg, Russian Federation). Candidate of Historical Sciences. Institute for the History of Material Culture of the Russian Academy of Sciences. Dvortsovaya Emb., 18, Saint Petersburg, 191186, Russian Federation E-mail: [email protected] Vladimir Semenov (Saint Petersburg, Russian Federation). Candidate of Historical Sciences. Institute for the History of Material Culture of the Russian Academy of Sciences. Dvortsovaya Emb., 18, Saint Petersburg, 191186, Russian Federation E-mail: [email protected] Varvara Busova (Moscow, Russian Federation). (Saint Petersburg, Russian Federation). Institute for the History of Material Culture of the Russian Academy of Sciences. Dvortsovaya Emb., 18, Saint Petersburg, 191186, Russian Federation E-mail: [email protected] Kharis Mustafin (Moscow, Russian Federation). Candidate of Technical Sciences. Moscow Institute of Physics and Technology. Institutsky Lane, 9, Dolgoprudny, 141701, Moscow Oblast, Russian Federation E-mail: [email protected] Irina Alborova (Moscow, Russian Federation). Candidate of Biological Sciences. Moscow Institute of Physics and Technology. Institutsky Lane, 9, Dolgoprudny, 141701, Moscow Oblast, Russian Federation E-mail: [email protected] Alina Matzvai (Moscow, Russian Federation). Moscow Institute of Physics and Technology. Institutsky Lane, 9, Dolgoprudny, 141701, Moscow Oblast, Russian Federation E-mail: [email protected]
Shopping Cart Items: 0 Cart Total: 0,00 € place your order
Price pdf version
student - 2,75 € individual - 3,00 € institutional - 7,00 €

Copyright В© 1999-2022. Stratum Publishing House

IMAGES
VIDEO
COMMENTS
Clinical Research Associate- Hybrid. Comprehensive Blood and Cancer Center. Hybrid work in Bakersfield, CA 93309. $80,000 - $120,000 a year. Full-time. Easily apply. Minimal knowledge of clinical research process, clinical trials and medical terminology. Experience: Candidate should have at least 7 - 8 years of clinical…. Active 11 days ago ·.
69 Clinical research associate ophthalmology jobs in United States. Wagner Kapoor Research Institute. Clinical Research Coordinator. Norfolk, VA. $19.00 - $22.00 Per Hour (Employer est.) Easy Apply. Completing data entry for all studies. Good Clinical Practice (GCP) certification.
Today's top 340 Clinical Research Associate Ophthalmology jobs in United States. Leverage your professional network, and get hired. New Clinical Research Associate Ophthalmology jobs added daily.
Clinical Research Associate- Hybrid. CBCC Bakersfield, CA. $80K to $120K Annually. Full-Time. Candidate should have at least 7 - 8 years of clinical research experience with at least 3 -4 years of monitoring experience for Oncology studies (Candidate with Oncology with ophthalmology ...
Omaha, NE. Be an early applicant. 1 month ago. Today's top 29 Clinical Research Associate Sr. Cra Ophthalmology jobs in United States. Leverage your professional network, and get hired. New ...
Previous experience as a Clinical Research Associate, preferably in ophthalmology or a related therapeutic area. Knowledge of ophthalmic diseases, treatments, and related medical terminology ...
The top companies hiring now for clinical research ophthalmology jobs in United States are Berkeley Eye Center, Boston Children's Hospital, Minnesota Eye Consultants, Infusion Associates, PLLC, Ophthalmology Specialists of Texas, UW Medicine Advancement, Pediatrix, Provident Research Inc, University of Washington Medicine, Edward-Elmhurst Health
We're 100% dedicated to ophthalmology. It's all we do. And with over 3,000 clinical ophthalmology projects completed, our teams are not just experts — they're THE experts. ... The preferred clinical research partner of ophthalmic innovators around the world. Preclinical. Retina, Glaucoma, Refractive & CED. Dry Eye & Ocular Surface.
Ophthalmology CRO Capabilities. Our medical, operations, and regulatory staff understand the complexities of ophthalmology trials from the perspective of the Sponsor, the clinical investigator, the scientific leader, and the reviewer at the regulatory agencies; we bring these perspectives to each clinical trial that we conduct to help keep trials running efficiently and according to protocol.
The Byers Eye Institute at Stanford University School of Medicine is seeking a Clinical Research Coordinator Associate to perform duties related to the coordination of clinical studies under the direction of Dr. Andrea Kossler, Associate Professor of Ophthalmology. Duties include coordination of moderately complex aspects of one or more ...
Clinical Research Associate Qualifications: Bachelor's degree in a related field (life sciences, nursing, etc.). Previous experience as a Clinical Research Associate, in ophthalmology
Collect and manage patient and laboratory data for clinical research projects. Manage research project databases, develop flow sheets and other study related documents, and complete study documents/case report forms. Ensure compliance with research protocols, and review and audit case report forms for completion and accuracy with source documents.
Vice Chair - Clinical Research. Associate Professor of Ophthalmology. Vice Chair, Clinical Research. Vitreoretinal Disease +1 919 684 4359. [email protected]. Associate Professor of Ophthalmology. Duke Eye Center, 2351 Erwin Road DUMC 3802, Durham, NC 27705. Mailing address. Duke Eye Center, DUMC 3802, Durham, NC 27710. Footer. Twitter;
Navbar Search Filter ... Mobile Microsite Search Term Search
Each has excelled in the areas of teaching, research and creative accomplishments, and service. ... Promotion to Associate Professor-Clinical: Ackermann, Wiebke, Anesthesiology Almaani, Salem, Internal Medicine ... Kerur, Nagaraj, Ophthalmology and Visual Sciences. College of Nursing. Promotion to Professor: O'Mathuna, Donal.
Title: Clinical Research Associate (Ophthalmology) Duration: 24 months . Location:Remote (however, candidate will need to be in specific states as they may occasionally have to go into the office.)
Zvenigorod's most famous sight is the Savvino-Storozhevsky Monastery, which was founded in 1398 by the monk Savva from the Troitse-Sergieva Lavra, at the invitation and with the support of Prince Yury Dmitrievich of Zvenigorod. Savva was later canonised as St Sabbas (Savva) of Storozhev. The monastery late flourished under the reign of Tsar ...
/PRNewswire-FirstCall/ -- Deere & Company (NYSE: DE) officially opened its new manufacturing and parts distribution facility south of Moscow today in...
Burial 5 was the most unique, it was found in a coffin made of a larch trunk, with a tightly closed lid. Due to the preservative properties of larch and lack of air access, the coffin contained a well-preserved mummy of a child with an accompanying set of grave goods. The interred individual retained the skin on his face and had a leather ...